|
All the elements arranged in the tabular form. The periodic table of element arranges all the chemical elements in increasing order of their atomic number, recurring chemical properties and electron configuration. , Seaborgium Valence Electrons, Dubnium Valence Electrons. Learn more about Valency here:- Hassium Valence Electrons It is an essential concept for understanding chemical bonding and the formation of compounds. In summary, the valency of elements varies across the periodic table based on their position and electron configuration. These elements tend to gain electrons to achieve a stable electron configuration. Group 16 elements, the chalcogens, have a valency of -2, and Group 15 elements, the pnictogens, have a valency of -3. They readily gain one electron to achieve a stable octet configuration. Transition metals, found in the middle of the periodic table, can exhibit multiple valencies due to their ability to lose different numbers of electrons depending on the specific reaction.Įlements in Group 17, known as halogens, have a valency of -1. Group 2 elements, the alkaline earth metals, have a valency of +2 as they tend to lose two electrons. This means they readily lose one electron to achieve a stable octet configuration. For example, elements in Group 1 of the periodic table, known as alkali metals, have a valency of +1. 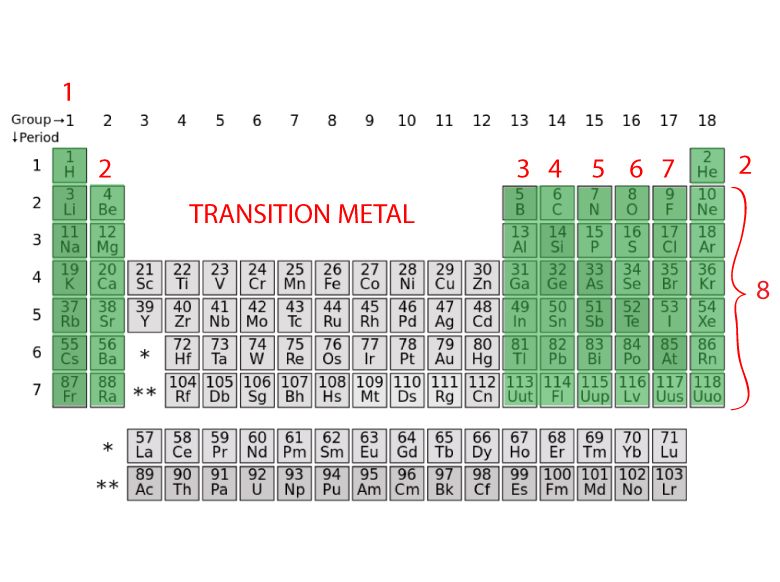
Explain the long form of the periodic table.Valency varies for different elements across the periodic table.How does the number of valence electrons vary on moving from left to right:(i) In the first period of the periodic table? (ii) In the second period of the periodic table?.Consider the following elements:Na, Ca, Al, K, Mg, Li (a) Which of these elements belong to the same period of the periodic table? (b) Which of these elements belong to the same group of the periodic table?.(a) What is the main characteristic of the last elements in the periods of the periodic table? What is the general name of such elements?(b) What is the number of elements in: (a) 1st period, and (b) 3rd period, of the modern periodic table?.Define the Periodic Table with elements name.Atomic number of a few elements are given below 10, 20, 7, 14(a) Identify the elements(b) Identify the Group number of these elements in the Periodic Table(c) Identify the Periods of these elements in the Periodic Table(d) What would be the electronic configuration for each of these elements?(e) Determine the valency of these elements.(a) What is the usual number of valence electrons and valency of group 18 elements of the periodic table?(b) What happens to the number of valence electrons in the atoms of elements as we go down in a group of the periodic table?. 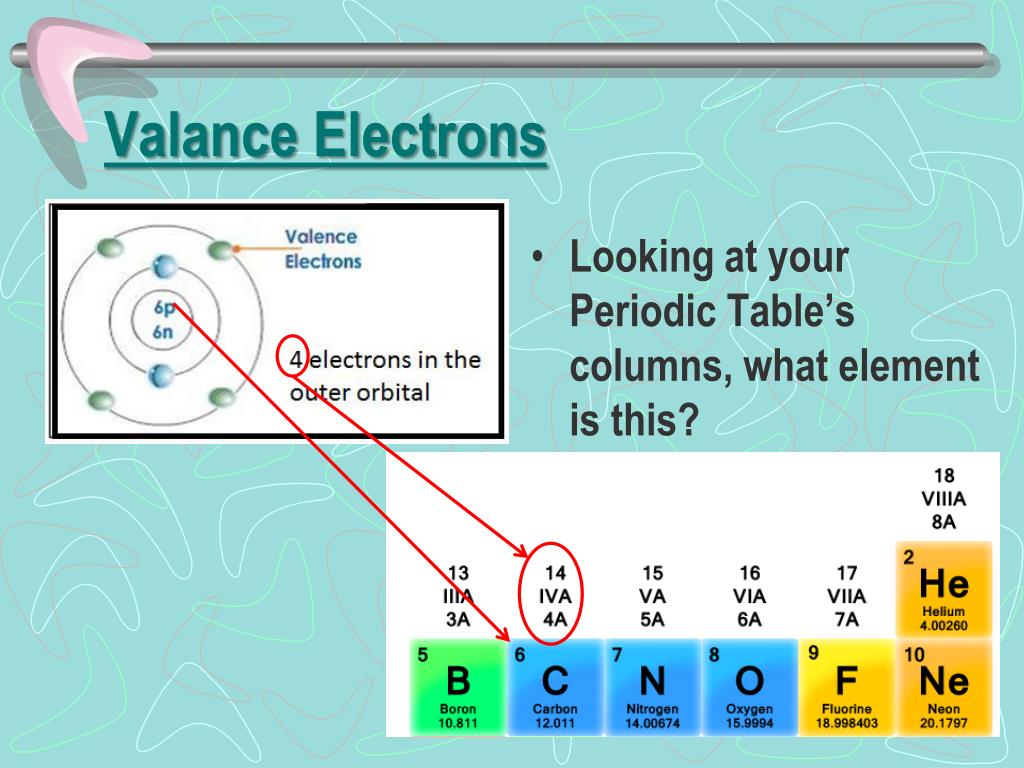
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
